Battery types used in portable and solar lighting
Rechargeable batteries are one of the most important components of portable and solar lights. Currently there are four popular types of rechargeable batteries used in portable and solar lighting – Ni-Cd, Ni-MH, Lead acid and Lithium-ion. We have created this extensive article to help you understand the structure, specifications, types, advantages and disadvantages of these batteries. Also, we have inspected and compared operation, prices, pros and cons of usage of each rechargeable battery in portable and solar lights.
At the end of the article we have made an informational table comparing specifications of lithium-ion, lead acid, Ni-MH and Ni-Cd rechargeable batteries and different useful visual charts that illustrate the comparison of four battery type specifications (capacity, specific energy, self-discharge rate, voltage, prices and other) . Click Here to go directly to comparison table and charts.
If you have any comments, questions or suggestions feel free to leave your message in the comment section under the article!
Ni-CD BATTERY
Ni-Cd (also known as Ni-Cad) is a type of secondary batteries that we are using (or were using) in many of our everyday portable appliances including flood lighting. “Secondary battery” means that Ni-Cd batteries are rechargeable.
Materials
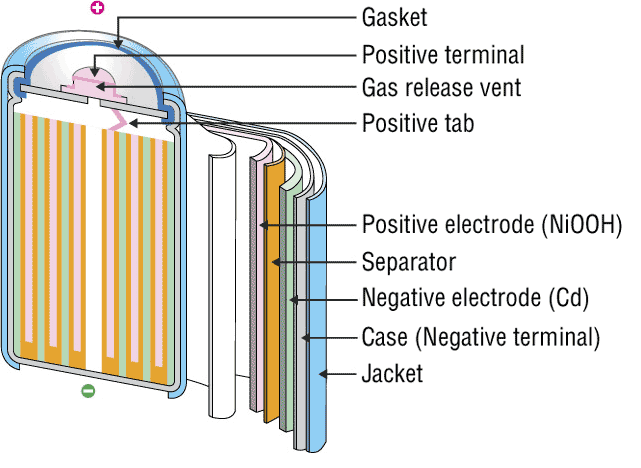
Ni-Cd battery is also called nickel-cadmium battery. As name suggests, it consists of nickel and cadmium, separator and alkaline. Ni-Cd battery consists of:
- Nickel oxide-hydroxide as positive electrodes;
- Metallic cadmium as negative electrodes;
- Potassium hydroxide alkaline electrolyte.
History
First wet-cell Ni-Cd batteries were invented in late 19th century. Ni-Cd batteries were by far the most popular type of batteries for smaller portable devices up till mid-1990s, when Ni-MH batteries took over the market. They were used in many portable devices, from small computers, toys, photo and video devices, power tools and others. The increasing popularity of Ni-MH (and later Li-ion) batteries made Ni-Cd batteries lose their market share. Ni-MH has better capacity (longer work time) at the same production price. Besides the growing popularity of Ni-MH batteries there were another thing that reduced consumption of Ni-Cd batteries in Europe – European Union Battery Directive. Ni-Cd batteries use highly toxic metal cadmium, which is poisonous to humans and can cause damage to the environment. The main problem with these Ni-Cd batteries is disposal of used batteries containing heavy metal cadmium. To address this issue, EU Battery Directive has greatly restricted usage of these batteries in electronic devices. Ni-Cd batteries are only allowed to be used in certain types of electric equipment.
Sizes and voltage
Cell voltage of Ni-Cd battery is 1.2V.
Ni-Cd batteries come in multiple different sizes with varying capacities. Basically, as Ni-Cd batteries were intended to replace older, primary (non-rechargeable) alkaline batteries, they come in same sizes as those alkaline batteries, so they can be used in the same portable devices. Ni-Cd batteries are made in sizes from AAA to D. Often Ni-Cd batteries are sold in packs of many (for example 4 battery pack), which have increased voltage and there are also other multi cell Ni-Cad batteries.
Charging and discharging
One of the main advantages of Ni-Cd battery is that it can maintain a steady voltage (1.2 volts) while discharging, even when it is close to being fully discharged. Other type of batteries like lead acid will lose the voltage when they are getting closer to discharge. Also, Ni-Cd batteries have low internal resistance, so they can charge and discharge a lot of power in very short period of time. Ni-Cd batteries can be fully charged in just approx. 1-2 hours and can be recharged approx. 400-500 times until they wear out and need to be changed.
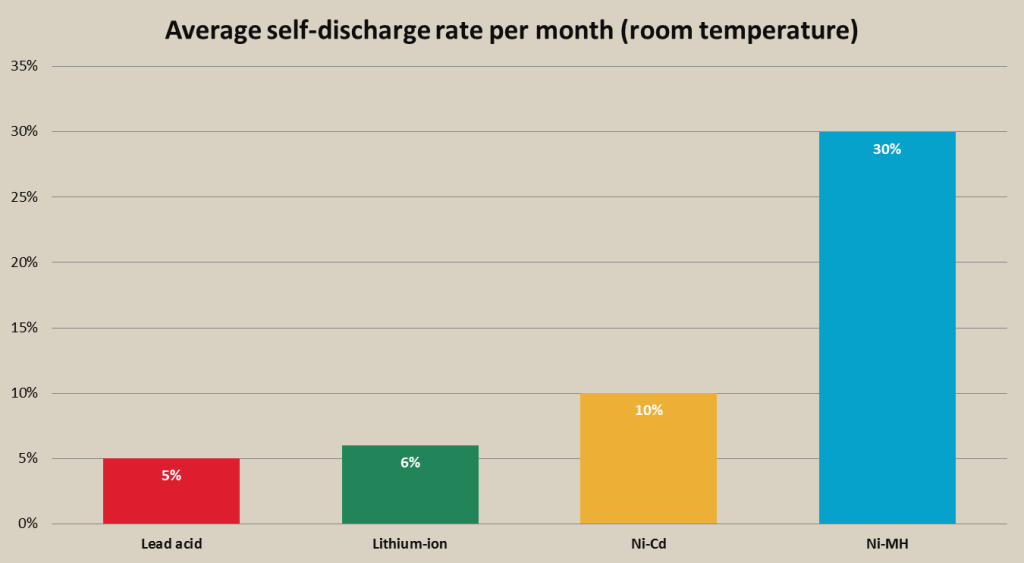
When not used, Ni-Cd battery will self-discharge about 10% of energy per month at normal temperatures, for example, if it’s left in a house on a shelf. However, if temperature in a place where battery is stored gets higher, the discharge rate per month can increase up to 20%. It is recommended to discharge the Ni-Cd battery before storage and store it in a cool and dry place.
Temperatures
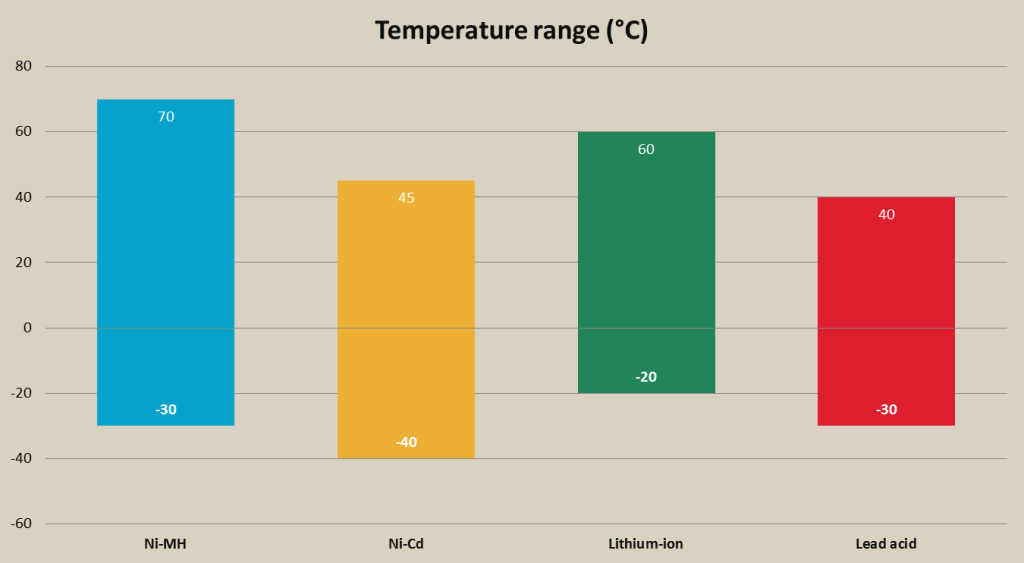
Ni-Cd battery can be used in quite large temperature range. The recommended temperature range to safely use Ni-Cd battery is from -20 to 45°C (-4 to 113°F), but these batteries can be used in temperatures as low as –40°C (-40°F).
Usage
Primarily, Ni-Cd batteries are used in smaller, portable electronic devices like remote controlled hobby toys, electric tools, TV and radio remote controls, flashlights, portable lights and others. Larger, specially made Ni-Cd batteries are used in larger electronic applications like emergency lighting and power work tools that requires large amount of power.
Ni-Cd Battery Advantages
- Ni-Cd batteries can be quickly charged and discharged;
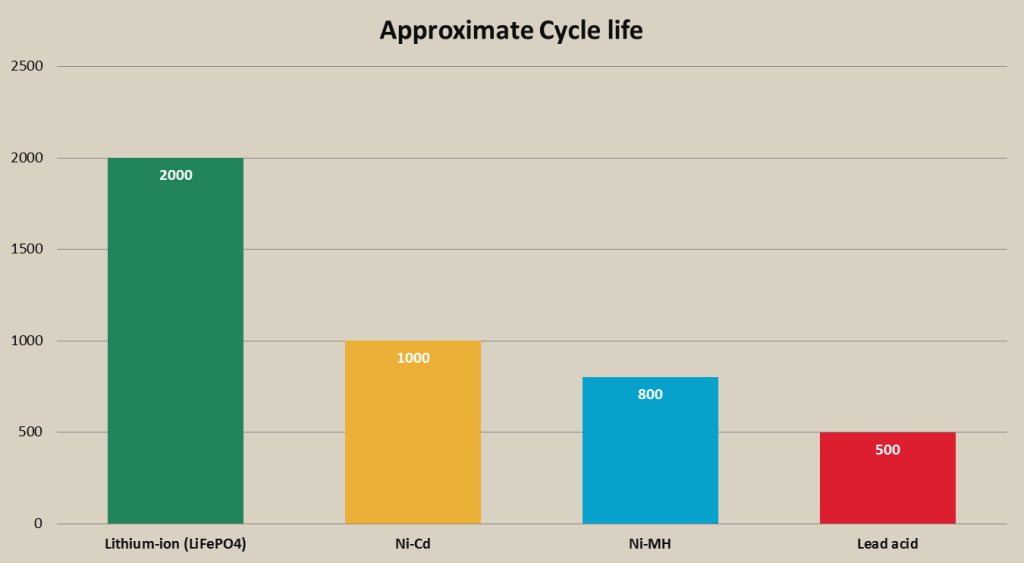
- Ni-Cd have better cycle life than Ni-MH, lead acid and some types of li-ion batteries; Ni-Cd batteries can provide even 3 times more charge/discharge cycles than Ni-MH batteries;
- Ni-Cd batteries can deliver their full capacity at discharge;
- Ni-Cd battery has good low temperature tolerance and can perform well in temperatures below -20°C (-4°F);
- Ni-Cd batteries are cheaper than Ni-MH and li-ion batteries, but more expensive than lead acid batteries.
Ni-Cd Battery Disadvantages
- Although Ni-Cd batteries have better self-discharge rate than Ni-MH batteries (approx. 10% a month compared to Ni-MH 30% a month), they still have higher self-discharge rate than lead acid and li-ion batteries.
- Ni-Cd batteries have significantly lower energy density than Ni-MH (approx. 2 times) and li-ion (approx. 3 times) batteries, but higher than lead acid batteries. Energy density is measured comparing watt hours to weight of the battery. Basically, Ni-Cd batteries can store much less energy in same weight battery than Ni-MH and li-ion batteries, but more energy than lead acid batteries, which have the lowest energy density from all of these rechargeable batteries.
- Ni-Cd batteries suffer from a “memory effect”. If battery gets discharged to the same level for multiple times, it will start to act like that is the lowest discharge level of the battery, so Ni-Cd battery will lose some of its capacity.
- Ni-Cd batteries use toxic metal cadmium, which is dangerous for environment. These batteries must be taken to a special disposal station that specializes in cadmium disposal, so empty NiCad batteries get properly recycled.
Ni-Cd battery usage in Portable and Solar lights
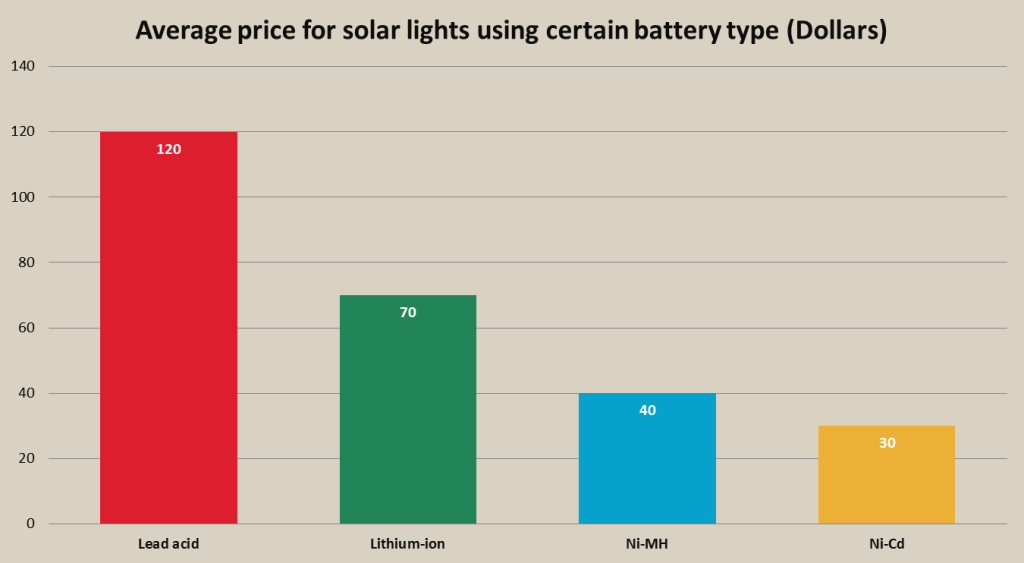
Ni-Cd batteries can be mostly found in low end portable and solar flood lights costing approx. 30 and less dollars. The closest rival to Ni-Cd batteries in low end flood lights are Ni-MH batteries, so it makes more sense comparing these two batteries, as li-ion and lead acid batteries are mostly used in mind high end flood lights. Low end flood lights are used in areas, where just “some sort of illumination” is needed, for example, front door light. Low price is the main advantage of such flood lights and as these lights are not intended to produce a lot of lumens, Ni-Cd batteries are often used in low end portable lighting because of their low cost. Ni-Cd batteries can be found in such manufacturer solar lamps as Reusable Revolution, Sunforce, LampLust.
There are three advantages of Ni-Cd batteries on portable and solar flood lights:
- Low price. Ni-Cd batteries cost less than Ni-MH batteries, so flood lights with Ni-Cd batteries usually are few bucks cheaper than those powered with Ni-MH.
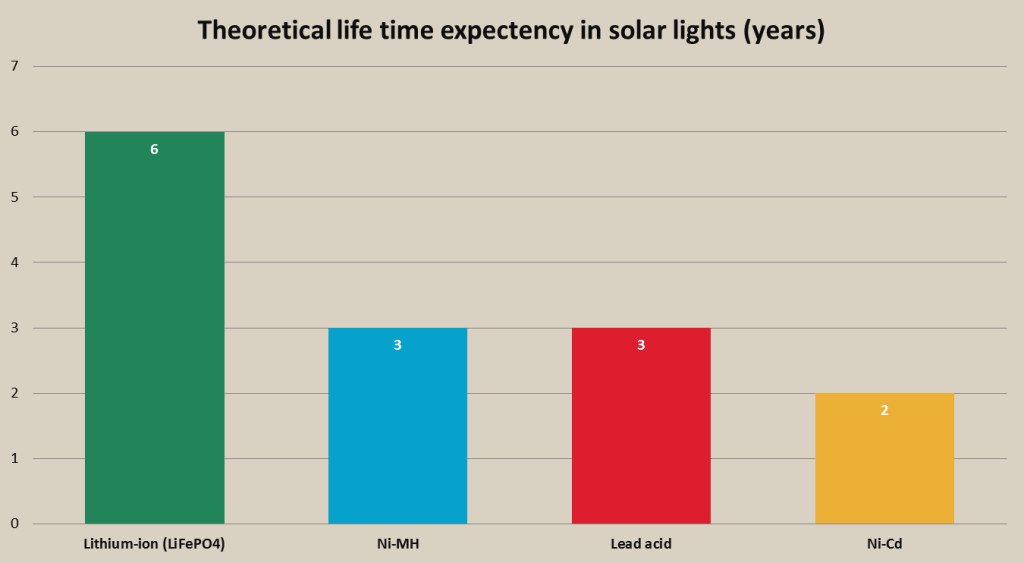
- Good cycle life. Ni-Cd batteries provide significantly more charge and discharge cycles than Ni-MH batteries. This means that in theory, Ni-Cd batteries in portable flood lights will need to be replaced after longer period of usage than Ni-MH batteries.
- Good low temperature tolerance. If you live in an area with very cold winters, where temperature often gets under -20°C (-4°F), flood lights with Ni-Cd batteries will be more stable and work better than Ni-MH batteries and li-ion batteries.
There are some serious disadvantages of Ni-Cd batteries, which make them much less used in portable lighting than Ni-MH and other type of rechargeable batteries:
- Low energy density. Ni-Cd batteries have much lower capacity than Ni-MH and li-ion batteries. In real life this means that Ni-Cd powered solar flood lights might not have enough capacity to store energy for full night illumination and light would stop working in a middle of the night.
- Memory effect. Development of memory effect can even more reduce the capacity of a Ni-Cd battery and decrease illumination time of a solar light.
- Toxic cadmium. Because of the toxic cadmium in Ni-Cd batteries, production and usage of these batteries have been restricted a lot in many countries, so you will see much less lamps using Ni-Cd than Ni-MH batteries.
Ni-MH BATTERY
Ni-MH (also known as nickel-metal hydride battery) is a type of secondary (rechargeable) battery that is mainly used in many smaller, portable consumer electronics. Ni-MH battery is very similar to Ni-Cd battery with its form factor and components, but instead of toxic cadmium used in Ni-Cd batteries, Ni-MH batteries have hydrogen absorbing alloy as negative electrode or anode. Also, Ni-MH batteries have surpassed Ni-Cd batteries and now are used in a lot wider range of electrical appliances. They have more than two times higher energy density than Ni-Cd batteries.
Materials
As mentioned, structure of Ni-MH battery is pretty similar to Ni-Cd battery, however, there are some differences in components used in Ni-MH batteries. Ni-MH battery consists of:
- Nickel hydroxide as positive electrodes;
- Hydrogen absorbing alloys (links) as negative electrodes;
- Potassium hydroxide alkaline electrolyte.
History
The beginnings of a Ni-MH battery does not go very far back in the history. This battery type was first patented in 1986 and became commercially available 3 years later in 1989. At that time market was ruled by Ni-Cd and alkaline batteries. But because of all advantages that Ni-MH battery could offer, it soon started to replace Ni-Cd batteries in portable consumer electronics and became the number one battery type used in these electronic devices. Also European Union Battery Directive restricted usage of Ni-Cd batteries in many electronic devices, because they contain toxic metal cadmium, and this allowed Ni-MH batteries to take Ni-Cd place in the market, as they have pretty similar features and form factor. Till about 2010 Ni-MH were dominating markets in many countries around the world. They were widely used not only in portable consumer electronics, but also in much larger industries. For example, Ni-MH batteries were widely used in hybrid cars. But in recent years, usage of Ni-MH batteries has decreased significantly because of technological development and production growth of lithium-ion batteries. At this time, Ni-MH batteries take less than 5% of all battery market.
Sizes and voltage
Cell voltage of Ni-MH battery is 1.2V and charging voltage is about 1.6V per cell.
The most common cell sizes of Ni-MH batteries are AAA and AA. C and D sized Ni-MH batteries are also available, but they are much more expensive. Because of that manufacturers often combine multiple AA cells in special adapters called battery packs to increase their voltage. Physical battery sizes of Ni-MH are very similar to Ni-Cd battery sizes.
Charging and discharging
One of the main advantages of Ni-MH battery is its high energy density. Ni-MH batteries have much larger energy density than Lead acid or Ni-Cd batteries. What this means is that Ni-MH battery can hold more energy than same sized Ni-Cd or Lead acid battery. Ni-MH battery can maintain a solid 1.2V approximately 70-80% of its discharge time. Ni-MH batteries cannot be completely discharged. A completely discharged cell can go into reverse polarity, which can damage the cell and cause problems in functionality of electrical device the cell is powering. Ni-MH battery cycle life can vary depending on the capacity of the battery. The average cycle life is from 500 cycles on high capacity batteries to almost 3000 cycles on lower capacity batteries. Ni-MH batteries can be rapid charged in 1 hour.
Probably the biggest disadvantage of Ni-MH battery is the high self-discharge rate. This means that if you are going to leave fully charged Ni-MH battery on a shelf, after few months (depending on the capacity of the cell) battery will have lost most of its charge. Usually, a Ni-MH battery can lose from 4-20% of its charge on first day and afterwards self-discharge rate drops to about 1% per day. This rate applies to room and lower temperatures, however, if temperature in the place, where Ni-MH battery is stored gets higher (over 40°C or 104°F) self-discharge rate of the battery is going to be much higher.
To over compensate this flaw, low self-discharge or LSD Ni-MH batteries were invented. They have much lower self-discharge rate, however these batteries also have lower capacity and their costs are little higher than regular Ni-MH cells.
Temperatures
Ni-MH batteries, same as Ni-Cd, have considerable temperature tolerance. The recommended temperature range to safely use Ni-MH battery is from approx. -30 to 70°C (-22 to 158°F). Maximum temperature range is always indicated by the manufacturer.
Usage
Ni-MH batteries replace older primary alkaline batteries and Ni-Cd batteries in small, portable consumer electronics. This means that they can be used practically in all the same electronic devices as both of those. The most common size of Ni-MH batteries are AA. Ni-MH batteries are mostly used in such devices as cameras, toothbrushes, radio controlled hobby toys, portable lighting and also in larger industries like medicine and car building. However, today we are seeing that Lithium-ion batteries are more and more taking over these markets reducing the demand for Ni-MH batteries.
Ni-MH Battery Advantages
- Ni-MH batteries have high capacity, more than two times higher than Ni-Cd and lead acid batteries. This means that Ni-MH battery will provide two times longer work time with one charge than Ni-Cd batteries;
- Ni-MH batteries are environmentally friendly. They do not contain any toxic substances that can be bad for the environment like cadmium in Ni-Cd batteries or lead in lead acid batteries. Ni-MH batteries can be recycled;
- Ni-MH batteries (at least those made more recently) do not suffer from a memory effect that Ni-Cd batteries have. This means that Ni-MH batteries can be charged and discharged at any rate.
Ni-MH Battery Disadvantages
- Ni-MH batteries cost more than Ni-Cd and lead acid batteries, but are cheaper than lithium-ion batteries;
- Ni-MH batteries have very high self-discharge rate, up to 3 times higher than Ni-Cd batteries and even more times higher than li-ion and lead acid batteries. Ni-MH battery will lose approximately third of its charge a month. This can increase based on the temperature, where batteries are stored;
- Ni-MH batteries are not suited for high charge and discharge rates as Ni-Cd batteries. They are suited to providing limited discharge currents. High discharge currents can reduce batteries life time;
- Ni-MH batteries suffer from risk of over-charging and over-discharging. Also Ni-MH batteries generate a lot of heat when charging. Special chargers designed for Ni-MH batteries must be used for charging.
Ni-MH battery usage in Portable and Solar lights
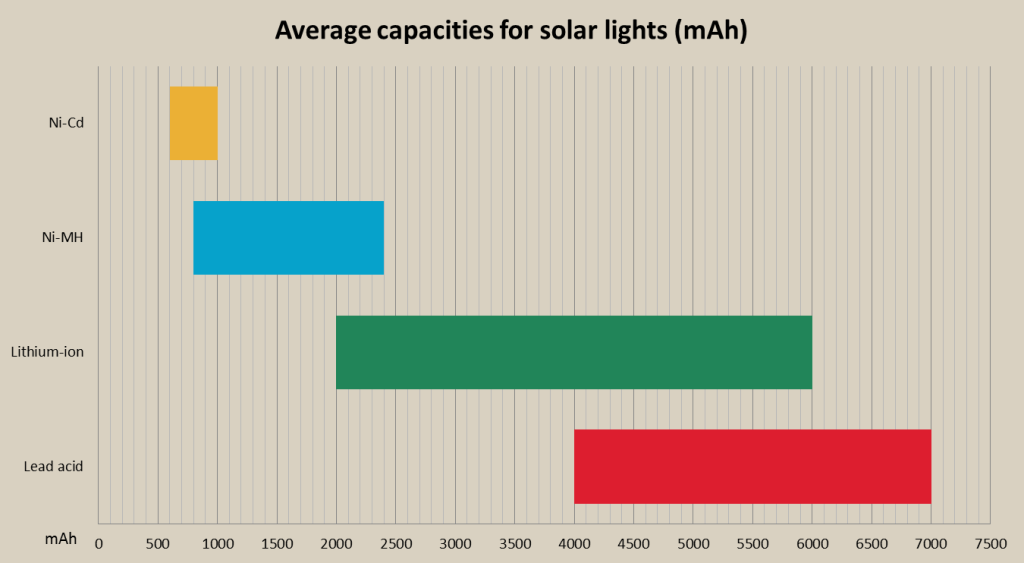
Ni-MH rechargeable batteries can be found in practically all range of solar flood lights form low end to high end lights, but mostly are used in low and mid-range lights. Flood lights using Ni-MH batteries cost from 20 dollars up to 100 and more dollars, but the average price is approx. 40 dollars. Ni-MH are much more frequently used in solar lights than Ni-Cd batteries, because they are more suited for lower voltage long duration discharge, instead of shorter time rapid discharge that Ni-Cd are capable of. Ni-MH flood lights are mostly used in household areas, starting from decoration and front door lights, to garden lights, shed lights, patio lights, security lights and many more. Ni-MH batteries are used in wider range of solar lights than Ni-Cd or lead acid batteries. Most portable lights come with Ni-MH batteries with approx. 2000mAh capacity in packs with 3-5 AA type Ni-MH batteries. Lights with Ni-MH batteries are produced by such manufacturers as Sunforce, Maxsa, Designers Edge, Solar Goes Green, All-Pro and others.
Ni-MH batteries have few advantages that make them so widely used in all ranges of portable and solar lights:
- High capacity. Ni-MH batteries have much higher energy density than Ni-Cd or lead acid batteries, which mean that they can store more energy in lighter and smaller battery, and for portable lights compactness of the fixture is an important factor. High capacity of the battery is especially useful for solar flood lights, as larger capacity means longer work times with one charge. While Ni-Cd batteries struggle to provide full night illumination for most solar lights even when they are equipped with a motion sensor, higher capacity Ni-MH lights can offer much longer work times and potentially could provide illumination from dusk till dawn.
- No memory effect. Ni-MH batteries do not develop a memory effect, so solar panel can charge battery to same rate for multiple times without any impacts to the capacity of the battery.
- Cheaper than li-ion. While Ni-MH batteries cost more than Ni-Cd battery, they are still cheaper than li-ion batteries, so they are more preferred in low end and mi-range lights to reduce the price of the lighting fixture.
There are also some disadvantages regarding Ni-MH battery usage in portable lighting:
- Lower cycle life. Ni-MH batteries have lower cycle life than Ni-Cd and li-ion batteries, which means that they will need to be replaced more frequently in solar lights than Ni-Cd or lithium batteries. However, replacement of these batteries usually is needed after more than 3 years of frequent usage.
- Over-charging and over-discharging. Ni-MH batteries have risk of over-charging and over-discharging, so lighting fixture must have specific charging mechanism that can deal with these issues.
- Self-discharge rate. Ni-MH batteries have high self-discharge rate, which means that if you leave your portable light with Ni-MH batteries on a shelf, it is going to lose significant amount of energy each day, and can lose all its charge in two months. So lighting fixture batteries must be checked and charged after fixture has not been used for longer period of time. This problem is not applicable to solar lights that are usually set in one place and are working and charging batteries each day.
LEAD ACID BATTERY
Lead acid battery is the oldest type of rechargeable battery and is also widely used today. Lead acid batteries can mostly be found in cars as starting batteries. Lead acid batteries are also used for high power portable flood lights.
There are two types of Lead Acid batteries – Starting and Deep Cycle batteries.
Starting batteries are used in places, where short and powerful energy outputs are needed, such as in automotive engines. These batteries have a lot more plates than deep cycle batteries. Also these plates are much thinner and have larger surface place so they can output higher current on discharge. Deep discharge cycles can heavily damage these batteries.
Deep cycle batteries are much better for long term usage and can handle a lot discharge cycles. Deep cycle batteries are not capable of producing as high and quick power bursts as starting batteries, but because of their thicker plates, these batteries are much better for frequent discharging. Deep cycle lead acid batteries are also used for portable flood lighting.
There are lead acid batteries that are combination of both starting and deep cycle batteries and can provide high starting current and also can withstand deep cycles. However, these batteries are not very common and are mostly used in specific applications.
Materials
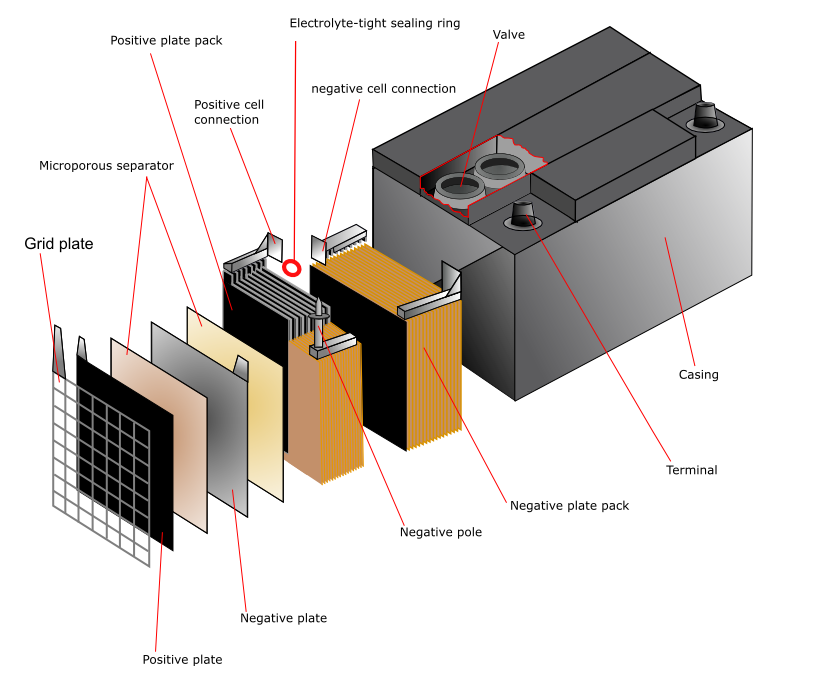
Lead acid battery consists of:
- Positive plate – lead dioxide paste;
- Negative plate – sponge lead;
- Separator – insulating material, is made from various materials, such as rubber, glass fiber, plastic and others;
- Electrolyte – sulfuric acid and water solution (35% and 65%);
- Casing – usually made from plastic.
Voltage is produced in reaction between positive plate (lead oxide), negative plate (lead) and electrolyte (sulfuric acid).
There are three types of lead acid batteries – Wet cell, Gel cell, AGM (Absorbed Glass Metal). Wet cell batteries are the oldest of these and are often called flooded batteries. In wet cell batteries a liquid electrolyte is used. This means that battery can mostly be used in an upright position, there is possibility that electrolyte can spill, they may require maintenance after some time. Wet cell batteries are mostly used as car starter batteries and their main advantage is low production cost.
Gel cell batteries use gel consistency electrolyte in opposite to liquid used in wet cell batteries. This gel is made from silica dust that gets added to electrolyte. Gel cell batteries are sealed, so they can be used in all orientations and also they do not sulfate as much as wet cell batteries. Gel cell batteries can withstand very deep discharge cycles. Gel cell same as AGM batteries cost significantly more than wet cell batteries so they are used in more specific applications. Both gel cell and AGM batteries are often known as VRLA or valve regulated lead acid batteries.
AGM or absorbent glass mat batteries uses glass microfiber mat separator between the battery plates. This fiberglass separator gets soaked into the electrolyte. AGM batteries are sealed and are considered maintenance free, so they do not require adding water. AGM, same as gel batteries can be used in all orientations, so they can be used in portable devices. AGM battery has longer lifespan, can produce higher discharge currents and is more resistant to lower temperatures. As mentioned above, AGM same as gel cell batteries are often called VRLA or valve regulated lead acid batteries.
Portable and solar flood lights are mostly equipped with one of the valve regulated lead acid batteries – gel cell or AGM.
History
Lead acid battery is the oldest type of secondary (rechargeable) battery and thanks to its low manufacturing cost it is still widely used today. These batteries were invented in 1859 by French physicist Gaston Plante. He laid the groundwork for the first rechargeable batteries and discovered the principle that a battery can be recharged again by passing a reverse current through the battery. This invention has not changed till today and lead acid batteries are still widely used as car starter batteries.
Before more than 40 years, the first sealed, valve regulated lead acid batteries were invented. Both absorbed glass metal and gel cell batteries later started to replace wet cell batteries in portable applications.
Today we can say that battery market is not going to change in the near future. Lead acid battery, thanks to the low manufacturing costs, is still the best solution for car batteries and will continue to be for at least few years.
Sizes and voltage
Lead acid batteries consist of multiple single cells. The approximate voltage of a single cell is 2.1 volts. Lead acid batteries come with different capacities and sizes. For example, one of the most common 12V lead acid batteries consists of 6 single cells. 6 cells will produce output voltage of 12.6V as voltage of single cell is 2.1 volts (The most common voltages of lead acid batteries are 2, 4, 6, 8 and 12 volts). Battery storage capacity depends on the size of the plates and amount of electrolyte. To increase the capacity of lead acid battery, multiple batteries can be connected together in parallel.
Charging and discharging
Cycle life of a deep cycle lead acid battery can vary dramatically based on depth of discharge and type of battery (wet cell, gel cell, AGM). The cycle durability of lead acid battery can vary from 500-3000 cycles and even more. For example, if valve regulated lead acid battery gets regularly discharged to 50% capacity it may provide approximately 1000 life cycles. However, this number can vary based on the type, quality and size of the battery.
The self-discharge rate of a lead acid battery depends on the storage temperature, same as with other types of rechargeable batteries. Lead acid batteries have self-discharge rate of approximately 5-20% a month. AGM lead acid batteries have very low self-discharge rate of approx. 2-3% a month.
Lead acid batteries should not be completely discharged, as it can damage the battery. As deeper a lead acid battery is going to be discharged, as less cycle life it is going to provide. For example, starting batteries can be fully discharged just few times, while some special valve-regulated deep cycle batteries can be fully discharged and provide even up to 1000 life cycles. It is also easy to determine state of charge of lead acid battery using several simple methods, so you can always tell the charge state of your battery.
Temperatures
The optimal temperature for lead acid battery usage is 20-25°C (68-77°F). Low and high temperatures are going to decrease lifespan of the battery. The approx. charging temperature range of lead acid battery is -30 to 40°C (-22 to 104°F), and approx. discharge rate -40 to 50°C (-40 to 122°F). If a battery is not fully charged the electrolyte may freeze at low temperatures.
Usage
Lead acid batteries are mostly used as starter batteries in transportation, for example in cars and motorcycles. Large lead acid batteries are used as backup power supplies for objects that need a permanent power source like hospitals, telecommunication buildings and other places. Valve regulated lead acid batteries are used in portable appliances as backup power sources, for example for computers and servers. Portable lead acid batteries can be found in many vehicles like electric bicycles and motorcycles. Valve regulated batteries are also used for high power portable flood and work lighting.
The popularity of lithium-Ion batteries is growing each year, however lead acid batteries are not going to be replaced by lithium-Ion batteries in the near future, because lead acid batteries are much cheaper to manufacture.
Lead acid Battery Advantages
- Lead acid batteries are very cheap to manufacture and have one of the lowest cost per watt hour of rechargeable batteries. This is one of the main reasons why lead acid batteries have been used for such a long time and will continue to be used in such huge industries as automotive;
- Lead acid batteries can produce very high discharge currents, so they are also used as automotive SLI or starting batteries;
- Lead acid batteries have very low self-discharge rate at about 5% a month and even lower for AGM batteries – approx. 2-3% a month;
- Newer AGM and Gel Cell batteries have good low temperature tolerance;
- Both wet and dry cell lead acid batteries do not develop a memory effect;
- Experienced and reliable technology that has been known for over 100 years.
Lead acid Battery Disadvantages
- Lead acid batteries usually come in a large size and are pretty heavy, which limits their usability in smaller portable devices;
- The main drawback of lead acid batteries are their low energy density, they have the lowest specific energy between Ni-Cd, Ni-MH and li-ion batteries. This means that lead acid batteries can store the least amount of energy based on the battery weight, which explains why lead acid batteries usually come in much larger form than other rechargeable batteries;
- Lead acid batteries need to be charged using slow charge and are not intended to be fast charged;
- Can have short cycle life (approx. 300-500 cycles) depending on the type of lead acid battery (Gel cell and AGM batteries provide longer cycle life than wet cell batteries)
- Lead acid batteries can’t be stored in a discharged state. Lead acid batteries can form a sulfation, if battery is stored fully or close to fully discharged state for longer period of time;
- Lead acid batteries are not friendly for the environment. They contain a chemical element – lead, which is dangerous for humans and the environment.
Lead acid battery usage in Portable and Solar lights
Lead acid batteries are mostly used in high end portable flood lights and solar flood lights that require a lot of power, and can also be found in some mid-range flood lights. Lead acid batteries are used to power flood lights and are much less used in other lamp types with much lower light output like spot lights. Lead acid batteries are used to power portable lights with up to thousand lumens and are often used to power light packs of 2, 4 or more lamps that are capable of producing multiple thousand lumen brightness and are often used in lighting systems. The closest competitor to lead acid battery is lithium-ion battery, especially lithium phosphate. Which battery type is used in which high power flood light usually depends on the manufacturer views. However, currently lead acid batteries are more popular than li-ion batteries in high power flood light and lighting systems because of their lower costs. As lead acid batteries are mostly used in high end flood lights which price range start from approx. 40 dollars up to 500 and more for large, high powered lighting systems. Lead acid batteries used in flood lights come with wide range of capacities and sizes, starting from 4Ah up to 50Ah and more. Flood lights using lead acid batteries are manufactured by such companies as Maxsa, Concept Enterprises, Solar Illuminations, Reusable Revolution, Solar Goes Green and others.
Advantages of lead acid batteries in portable and solar flood lights:
- Cheap cost. The biggest advantage of lead acid batteries are their cheap manufacturing costs and high power they can produce, which is one of the main reasons, why many manufacturers chose to use lead acid batteries in their flood lights instead of lithium-ion batteries. If more capacity is needed, multiple batteries can be used together to provide longer work hours.
- Low self-discharge rate. The low self-discharge rate of lead acid battery is great plus for portable flood lights and emergency lighting. Lead acid battery self-discharge only 2-5% a month, so flood lights with lead acid batteries can be stored unused for relatively long time.
- Good low temperature tolerance. Flood lights with lead acid batteries can be used in lower temperature range, over -30°C (-22°F) without any problems. This is an advantage for those, who live in areas with colder climate and need to use a flood light in winters.
- Valve regulated lead acid lights do not require practically any maintenance, which is useful for solar flood lights, and these batteries can be used in any orientation, which is good for portable flood lights.
Disadvantages of lead acid batteries in portable and solar flood lights:
- Low energy density. Low energy density means that lighting fixtures using lead acid battery must be made in larger size or need to have an external battery in order to provide the same capacity as li-ion or Ni-MH batteries.
- Limited usage. The large size and weight of lead acid battery limits its usage in smaller portable and solar lights like, for example, solar spot lights. This is the main reason why you won’t find low end portable and solar flood lights with low light output powered by lead acid batteries.
- Short cycle life. The low cycle life of lead acid battery means that you will need to change battery of your solar flood light after few years of usage. Cycle life of lead acid battery is visibly shorter than li-phosphate batteries. The good side on this is that Gel Cell and AGM batteries have much improved life cycles, which can even go up to 2000 cycles.
LITHIUM-ION BATTERIES
Lithium-ion (also known as li-ion) currently is the most common rechargeable battery type. Older lithium batteries used lithium metal, but due to its instability and safety issues lithium ions were used instead. Lithium-ion batteries offer number of advantages over other type of batteries such as high energy density and low maintenance, which makes them the best solution for widely used portable applications such as laptops, smartphones, digital cameras and others.
Lithium-ion batteries have various types that are used for different applications. The most common types are Li-cobalt, Li-manganese, Li-phosphate and NMC (lithium nickel manganese cobalt oxide). Each of these Lithium-ion batteries uses different cathode materials, so each of them has different pros and cons. For example, li-cobalt batteries are the most popular ones and are used in laptops, smartphones and similar devices, while li-phosphate batteries are used in electric vehicles and portable lighting.
Materials
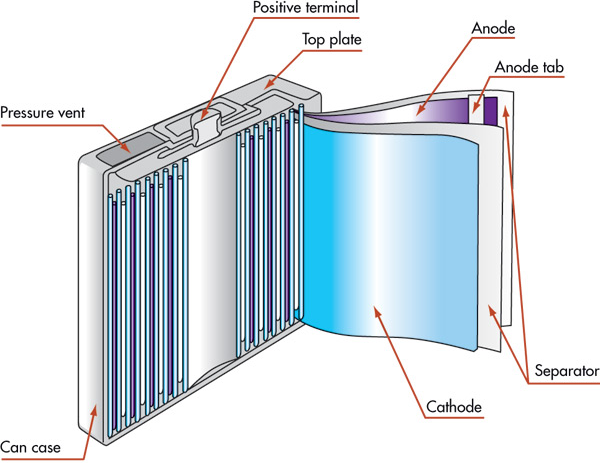
In a li-ion battery when discharging ions travel from negative electrode to positive and in reverse when charging. Lithium-ion battery consists of:
- Pure lithium metal oxide as cathode, more specifically
- lithium cobalt oxide for li-cobalt batteries;
- spinel or lithium manganese oxide for li-manganese batteries;
- lithium iron phosphate for li-phosphate batteries;
- Lithium Nickel Manganese Cobalt Oxide for NMC batteries.
- Porous carbon, more specifically graphite as anode;
- Lithium salts combined with organic solvent as electrolyte;
- Thin sheets of microporous plastic usually polypropylene or polyethylene as separator.
Lithium Cobalt Oxide batteries
Lithium cobalt oxide (LiCoO2) batteries are mostly used in common portable electronics like laptops, tablets, smartphones, digital cameras and other. Li-cobalt batteries have high specific energy density, which means that they can store more energy based on the mass of the battery. However, they need special monitor systems to ensure proper safety, they have lower specific power and lithium-cobalt batteries have shorter life and cycle span than competing types of lithium-ion batteries.
Lithium Manganese Oxide batteries
Lithium manganese oxide (LiMn2O4) batteries have high specific power and they are used for devices that require high power discharge, for example power tools and electric vehicles. These batteries have lower internal resistance, so they can charge and discharge at higher rates. Li-manganese batteries also are safer and can provide better life span than li-cobalt batteries. However, li-manganese batteries have lower capacity than li-cobalt batteries.
Lithium Iron Phosphate batteries
Lithium Iron Phosphate (LiFePO4) batteries are used in variety of electronic products, starting from portable consumer electronics to electric vehicles. Li-phosphate batteries are also the most common type of lithium-ion batteries used in portable flood lights. These batteries are safer than Li-cobalt and Li-manganese batteries, they have better life span and can produce high currents (more powerful than li-cobalt). However, li-phosphate batteries have one of the lowest capacities (lower than li-manganese) of all lithium-ion battery types.
Lithium Nickel Manganese Cobalt Oxide (NMC)
Lithium Nickel Manganese Cobalt Oxide (LiNiMnCoO2) or NMC batteries are mostly used in electric transportation applications such as electric cars, bikes, scooters, bicycles and other. NMC batteries use nickel manganese and cobalt combination as a cathode material. NMC batteries provide good specific power and high capacity, they have good life span and temperature tolerance, and these batteries have decent safety compared to li-cobalt batteries. NMC batteries try to combine the best features of other lithium-ion batteries.
History
The first experiments and developments of lithium battery started in early 20th century, however, the first lithium batteries for commercial purposes were made just in 1970s. These were primary batteries (non-rechargeable) and metallic lithium was used as anode material instead of lithium ions. Metallic lithium is reactive material, so lithium metal batteries caused serious safety issues. Because of this, scientists started to use non-metallic lithium ions to produce safer lithium batteries.
The first commercially available lithium-ion battery was introduced by Sony in 1991. Since then the market of Li-ion batteries has developed and grown rapidly and today lithium-ion batteries provide much better performance and higher capacity. Also an important factor in progression of lithium-ion battery is the reduction of its price. The first Li-ion batteries were very expensive to manufacture, a lot more expensive than Ni-Cd or Ni-MH and lead acid batteries, but progression of technologies and materials used in these batteries allowed to improve their performance, while at the same time reducing manufacturing price of Li-ion batteries.
In 1996 the first lithium iron phosphate batteries were introduced that are being used in different portable applications, including portable lights.
Today, lithium-ion batteries are used in variety of industries starting from consumer electronics (smartphones, laptops, tablets etc.) to automotive industry (electric car batteries). Lithium-ion battery is certainly the battery of the future and new materials and technologies of lithium-ion batteries are introduced each year that further improves capacity, power, life span and safety of these batteries.
Sizes and voltage
The nominal cell voltages of Li-ion batteries are:
- Li-cobalt (LiCoO2) – 3.6V
- Li-manganese (LiMn2O4) – 3.8V
- Li-phosphate (LiFePO4) – 3.2/3.3V
- NMC (LiNiMnCoO2) – 3.6/3.7V
Lithium-ion batteries come in various shapes and sizes, starting from small, coin shaped cells to large battery packs used in electric vehicles. Lithium batteries are available in same common AAA and AA sizes, however, usually lithium-ion cells come in packs of many to form a larger battery (usually prismatic shaped) that can be used for different electric appliances, including portable lighting. Lithium-ion batteries used in portable devices such as laptops, smartphones and tablets come in many variations of shapes and sizes, and are specially designed for different manufacturers and products.
Charging and discharging
Based on the type of lithium-ion battery, charging and discharging rates, self-discharge and charging temperatures can vary significantly. When lithium-ion battery discharges, ions travel from negative to positive electrode, and in reverse direction when charging. To charge a lithium-ion cell, a same polarity higher voltage gets applied to the battery, which makes the lithium ions move from positive to negative electrode.
Lithium-ion batteries are better charged in lower than higher temperatures. When battery gets charged in large heat, it can reduce batteries life span or even damage the battery. The accepted temperature range for charging Li-ion battery depends on the type of the battery, but usually for consumer electronics it is from 0 to 45°C (32 to 113°F). It takes about an hour to fully charge most common Li-ion batteries used in consumer electronics.
Self-discharge rates of lithium-ion batteries can differ based on the type of the battery, but average rate is approximately 2-8% per month. These rates can vary depending on different factors like age of the battery and temperature in which battery is stored. Older lithium-ion batteries will have larger self-discharge rate. Lithium batteries need to be stored in a cool and dry environment. If the battery is left in an area with temperature of 40°C (104°F) and larger, the self-discharge rate can rise more than 10 times, meaning that the battery is going to self-discharge at rate of 15-20% a month.
The average lifespan of lithium-ion battery is from 500 to 2000 cycles. Again, life span heavily depends on the type of lithium-ion battery and conditions the battery is used. For example, li-cobalt batteries have almost two times shorter lifespan than li-phosphate batteries. The lifespan of a lithium-ion battery will be longer, if the battery does not get fully discharged.
Temperatures
The recommended charging temperature range for Li-ion batteries is from 0 to 40°C (32 to 104°F) and discharging temperature range from -20 to 60°C (-4 to 140°F), although going close to highest and lowest temperature margins can impact lifespan of the battery. Charging and discharging temperatures can vary depending on the type of li-ion battery, for example, li-phosphate batteries that are used in portable flood lights will have greater operation and charging temperature range than li-cobalt batteries used in laptops and smartphones.
Usage
Lithium-ion batteries are one the most popular rechargeable batteries that are used in different types of electronic devices. Thanks to their advantages like high energy density, light weight and low maintenance, lithium-ion batteries are superior than Ni-Cd, Ni-MH and Lead Acid batteries in multiple ways, which makes them preferred rechargeable battery type in most new consumer electronics, power tools and electric vehicle industries.
Each of lithium-ion battery types (li-cobalt, li-manganese, li-phosphate, NMC and other, newer technologies) has different features and advantages, which makes them more suited for certain applications. For example, li-cobalt batteries, because of their capacity are used as laptop, smartphone, tablet, digital camera batteries, li-manganese batteries thanks to their high power are used in power tools such as drills, saws and others, li-phosphate batteries thanks to their long cycle life, wider operation temperature range and safety are used for electric vehicles as well as portable lighting, and NMC batteries, thanks to their overall performance are the preferred battery type for electric vehicles such as electric cars.
Currently lithium-ion battery technology is improving and new materials are being used in these batteries to improve their capacity, life span, power and safety, so we are probably going to see more lithium-ion batteries replacing other types of batteries in larger industries like, for example lead acid batteries in automotive industry. Growing popularity of electric car production and increasing demand for portable devices with longer battery life pushes research of lithium-ion batteries further, which results in development of new types of lithium-ion batteries each year. However, lithium-ion batteries have been around for a while and their potential is not limitless, so many companies and scientists are working to develop other battery concepts beyond li-ion batteries. But currently none of these experiments are close to being used in commercial applications and consumer electronics, so for few years now we are going to use lithium-ion as our primary rechargeable battery.
Lithium-ion Battery Advantages
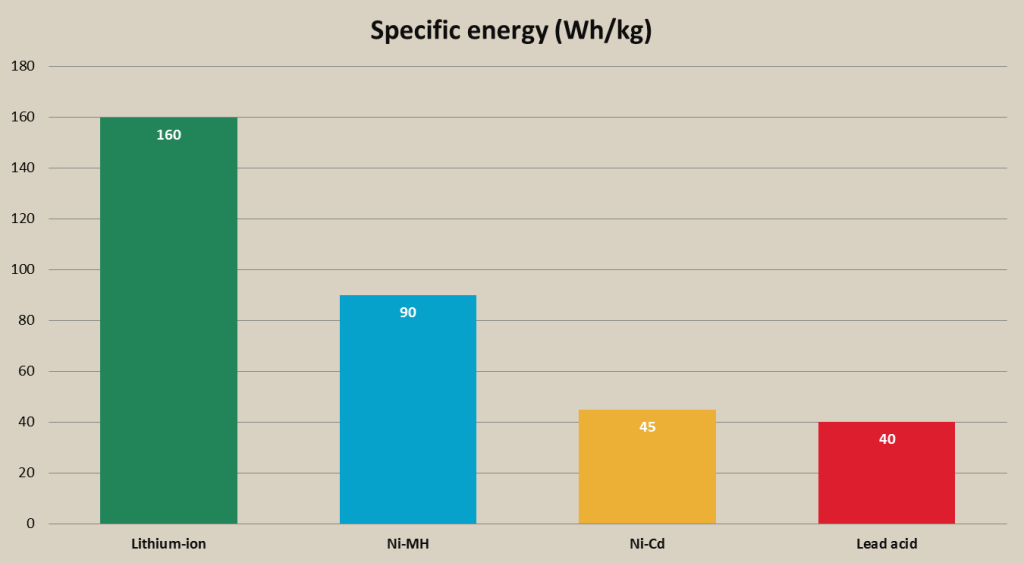
- Lithium-ion batteries have high specific energy, which makes these batteries the best option for smaller portable devices like smartphones, laptops and others. Their energy density is notably higher than Ni-MH, twice as high as Ni-Cd and more than three times higher than lead acid batteries. The highest specific energy of the lithium-ion batteries has li-cobalt battery;
- Li-ion batteries have low self-discharge rate, lower than Ni-Cd and Ni-MH batteries. The self-discharge rate can vary from 2%-8% based on the type of li-ion battery and temperature in which the battery is stored;
- Li-ion batteries do not develop a memory effect;
- Li-ion batteries are practically maintenance free, so they can be left in an electric device without a need for any maintenance, which is especially useful for solar lighting;
- Li-phosphate batteries that are used in such applications as portable and solar lightings have long cycle life (up to 2000 cycles). Other popular li-ion battery types like li-cobalt and li-manganese have shorter cycle life;
- Li-ion batteries are safe for environment and does not contain any highly toxic substances;
- Because of high energy density, li-ion batteries have light weight and can be made in small size, which is useful for compact portable devices and electric appliances, where low weight of a product is important;
- Li-ion batteries have multiple types like li-cobalt, li-manganese, li-phosphate, NMC and others that each have different advantages and each are used in specific industries;
- Li-ion batteries allow fast charging to full capacity;
- Safety – li-phosphate batteries used in portable lighting have very good thermal and chemical stability.
Lithium-ion Battery Disadvantages
- Li-ion batteries have high manufacturing costs resulting in high sale prices. Currently their price is higher than Ni-Cd, Ni-MH and lead acid battery price. The price is getting lower each year, but Li-ion batteries still cost more than competing rechargeable batteries;
- Li-ion batteries requires protection circuits to limit voltage and currents and ensure better safety;
- Some Li-ion batteries like li-phosphate provide low discharge rates;
- Li-ion batteries suffer from aging even when they are not in use;
- Li-ion battery can lose capacity and cycle life when stored in higher temperatures (over 30°C or 86°F) for longer period of time.
Lithium-ion battery usage in Portable and Solar lights
Lithium-ion batteries, same as Ni-MH, are used in wide range of flood lights, starting from cheaper, low end lights up to powerful, high end flood lights, but can be mostly found in mid-range and high end portable and solar lamps. Because of their large capacity, lithium-ion batteries can be made small enough for compact, portable and solar lights, but at the same time are also widely used in mid-range and high end lights. As lithium-ion batteries are used in such wide range of lights, they are directly competing with every other rechargeable battery type.
The most common lithium-ion battery type used in portable and solar lights is lithium phosphate (LiFePO4). These batteries provide some vital advantages over other rechargeable batteries, including other lithium batteries such as great cycle life (up to 2000 charge/discharge cycles), are very safe, can produce high currents, which is useful for powerful flood lighting systems, and have pretty large specific energy (approx. 120 Wh/kg), which although is smaller than li-cobalt or li-manganese, still is larger than other battery types. The price of a portable or solar light with lithium-ion battery starts from 30 dollars and goes over multiple hundred dollars for powerful lighting systems. Li-ion batteries for portable lights and solar lights come with wide range of battery capacities, starting from 2Ah to 30Ah and more for high power lighting systems. Portable and solar lights powered by lithium-ion batteries are manufactured by companies as eLEDing, Solar Goes Green, MicroSolar, Ivation, Lighting EVER, Solar Illuminations, Northen Twilight, Reusable Revolution, XEPA and others.
There are multiple advantages of li-ion battery usage in portable and solar lighting:
- High specific energy. The main advantage of lithium-ion battery usage in lighting is its high specific energy. This allows producing batteries with same capacity as other rechargeable batteries, but much more lighter and compact. This is why lithium-ion batteries are used not only in high powered flood lights but also in smaller, portable lamps like spot lights.
- Low self-discharge rate. The low self-discharge rate of approx. 2-8% per month is an advantage for portable lights and emergency lighting, because lighting fixtures do not need to be recharged after they have been stored unused for a few months.
- Maintenance free. Li-ion batteries are practically maintenance free and do not develop any memory effect, which is especially useful for solar lights, as they can be installed and left working without need of any maintenance for a long time.
- Good cycle life and safety of a li-phosphate battery. Li-phosphate is the most used lithium battery type for solar lighting, because it offers very good cycle life (over 2000 cycles) and is very safe to use even in higher and lower temperatures.
There are few disadvantages of li-ion battery usage in portable and solar lighting:
- High costs. High manufacturing costs of a lithium-ion battery is their main drawback. Usage of a li-ion battery in a flood or spot light is more expensive then usage of other rechargeable batteries like Ni-MH or lead acid, and this results in a higher total price of the lighting fixture. However, the cost of low and mid-range lights with li-ion batteries is not very much higher than price of similar lights with Ni-MH batteries. In high end flood lights and lighting systems, lead acid batteries are significantly cheaper to use than lithium-ion batteries, so many manufacturers are still preferring lead acid batteries over lithium-ion in such lights.
- Lithium-ion batteries still suffer from aging of the battery even if they are not being used, which means that the battery loses some of its capacity over time. Still, li-ion batteries can provide the same or even better cycle life than Ni-MH and VRLA batteries.
- Low temperature intolerance. Low charging and operation temperatures (-20°C or -4°F and less) can impact cycle life of a lithium-ion battery. Lithium phosphate batteries have better low temperature tolerance and can work in temperatures below -20°C (-4°F) in opposite to other lithium-ion batteries.
COMPARISON OF Ni-Cd, Ni-MH, LEAD ACID, LI-ION BATTERIES
For those, who do not like to read through lots of text we have created some visual materials comparing these four rechargeable battery types. We have done some general comparison as well as gathered data from over 100 portable and solar lights from the most popular lighting manufacturers and compared different battery type usage in portable and solar lights. Also, these resources were used to collect more in-depth data about rechargeable battery specifications:
If using any of our visual materials, please provide link back to our website www.ledwatcher.com
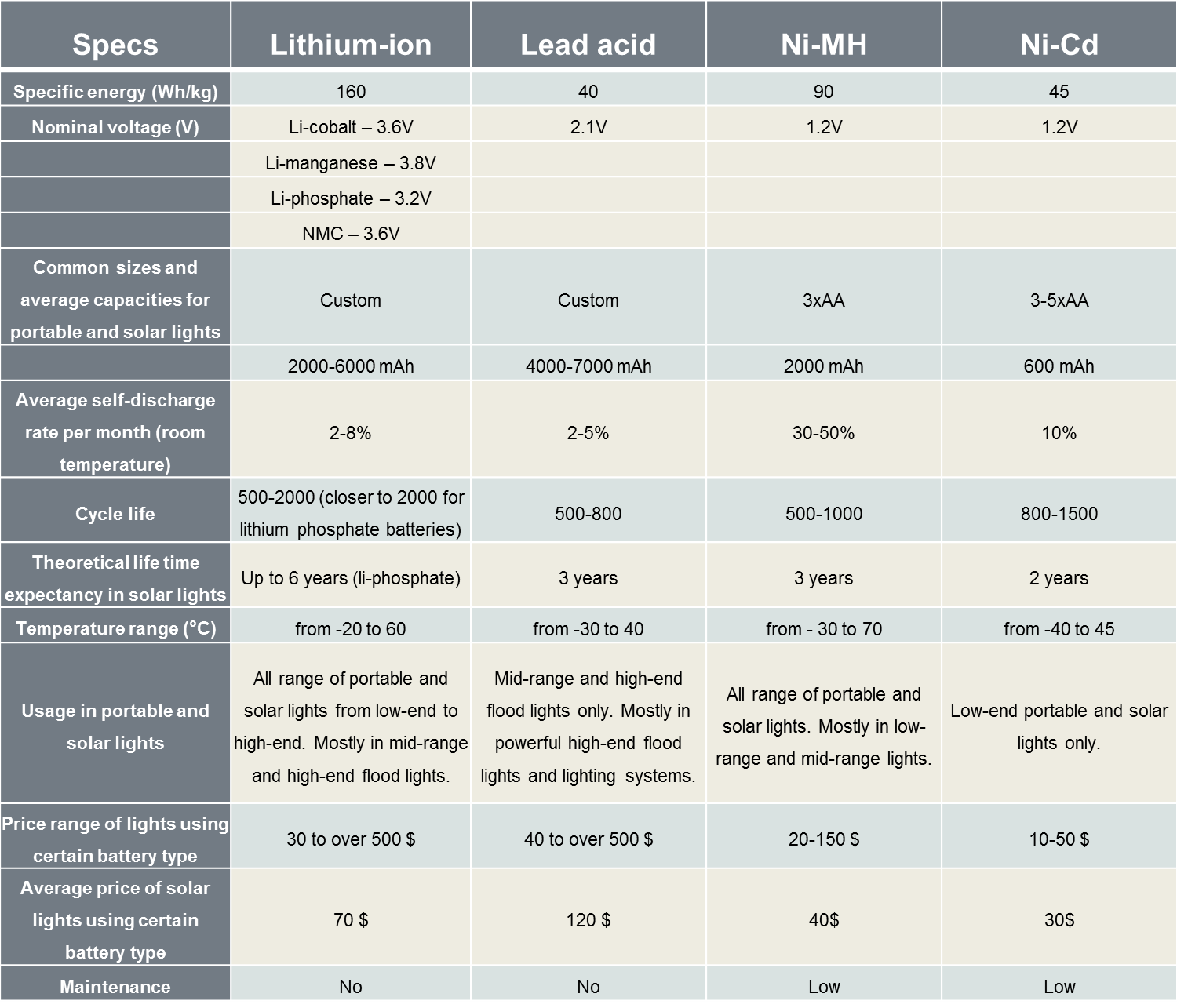
An informational table comparing specifications of lithium-ion, lead acid, Ni-MH and Ni-Cd rechargeable batteries
Rechargeable battery specific energy comparison chart
Rechargeable battery nominal voltage chart
Rechargeable battery average capacities for solar lights chart
Rechargeable battery average self discharge rate per month comparison chart
Rechargeable battery approximate cycle life comparison chart
Rechargeable battery theoretical lifetime expectancy in solar lighting chart
Rechargeable battery temperature range comparison chart
















Thanks for writing such a helpful info about different types of batteries, I have founded great stuff on this blog.
Handful of information..thanks
thanks for helpful information about batteries
Do any of these come in C size? I have an inexpensive unit from Menards that is loosing charge quickly and the 3 batteries that came with it appear to be regular c batteries. They do not even say rechargeable on them.
You should look into the EBL Rechargeable C-size Batteries.